Electrons are negatively charged subatomic particles that play a central function within the construction of atoms and the habits of matter. Though electrons have little or no mass in comparison with protons and neutrons, they occupy most of an atom’s quantity and decide how atoms work together with each other. From chemical bonding to the properties of parts, electrons are important to understanding chemistry and atomic construction.
Electrons are drawn to the positively charged nucleus of an atom and exist in particular areas related to outlined power ranges. These areas are organized into shells, sub-shells, and orbitals, every describing the power, place, and likelihood of discovering an electron.
What Are Electrons?
An electron is a subatomic particle with a destructive electrical cost of –1. Not like protons and neutrons, that are discovered within the nucleus, electrons transfer across the nucleus in areas of area known as electron clouds.
Though electrons have negligible mass—about 1/2000 the mass of a proton—they’re critically necessary. Their association determines whether or not an atom is secure, the way it reacts with different atoms, and what chemical bonds it might type.
Electrons Orbit the Nucleus
Electrons are discovered exterior the nucleus in discrete areas related to power ranges, also known as electron shells.
Electron Shells and Power Ranges
-
Electrons nearer to the nucleus have decrease power
-
Electrons farther from the nucleus have greater power
-
As distance from the nucleus will increase, the power of the electron will increase
The innermost shell can maintain solely a small variety of electrons, whereas outer shells have extra space and might maintain extra electrons. This construction explains why atoms can have many electrons with out collapsing inward.
Sub-Shells and Orbitals
Electron shells are additional divided into sub-shells, which describe power ranges extra exactly. Every sub-shell incorporates a number of orbitals.
What Is an Orbital?
An orbital is just not a set path like a planet’s orbit. As an alternative, it’s a area of likelihood the place an electron is most definitely to be discovered. Orbitals come in several shapes and orientations, reflecting the complicated habits of electrons.
Collectively, shells, sub-shells, and orbitals create a structured system that governs how electrons are distributed across the nucleus.
Electron Power and Distance from the Nucleus
The power of an electron is instantly associated to its distance from the nucleus:
-
Low power electrons are discovered nearer to the nucleus
-
Excessive power electrons occupy outer shells
-
Outer shells have extra room and might maintain extra electrons
Due to this association, electrons fill the bottom out there power ranges first earlier than occupying greater ones. This sample is important for understanding atomic stability and reactivity.
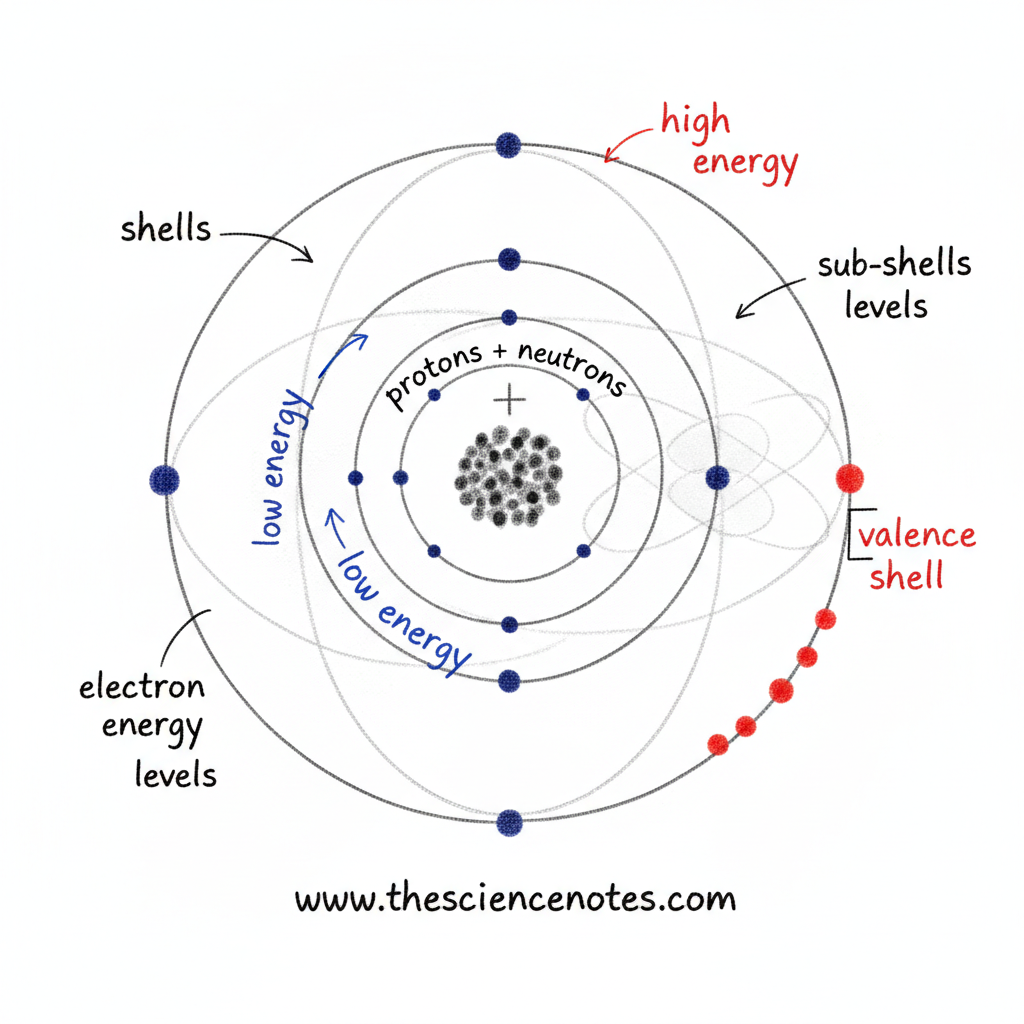
Valence Electrons and Chemical Properties
The electrons within the outermost shell of an atom are known as valence electrons. These electrons are particularly necessary as a result of they’re concerned in chemical bonding.
Why Valence Electrons Matter
Valence electrons decide:
-
The reactivity of a component
-
The varieties of chemical bonds it might type
-
The bodily and chemical properties of the ingredient
Atoms have a tendency to achieve, lose, or share valence electrons with a purpose to obtain a extra secure electron configuration.
Ionic and Covalent Bonds
Valence electrons permit atoms to type bonds in two primary methods:
Ionic Bonds
-
Kind when electrons are transferred from one atom to a different
-
One atom turns into positively charged, the opposite negatively charged
-
Widespread in salts and ionic compounds
Covalent Bonds
These bonding behaviors clarify how atoms mix to type the huge number of substances present in nature.
Discovering the Electron
The electron was the first subatomic particle to be found, marking a serious turning level in atomic concept.
J. J. Thomson and Cathode Ray Tubes
Within the late Eighteen Nineties, physicist J. J. Thomson carried out experiments utilizing cathode ray tubes—glass tubes with electrodes linked to an influence supply.
When electrical energy was utilized:
-
A beam of particles traveled from the destructive electrode (cathode) to the constructive electrode (anode)
-
A phosphor-coated display glowed when struck by the beam
This beam was referred to as a cathode ray.
Proof of Unfavourable Cost
Thomson handed the cathode ray between two charged steel plates:
-
One positively charged
-
One negatively charged
The ray bent towards the positively charged plate and away from the negatively charged one. Since reverse fees appeal to and like fees repel, this demonstrated that the particles within the ray carried a destructive cost.
Measuring Electron Mass
Additional experiments allowed Thomson to calculate the mass-to-charge ratio of the cathode ray particles. The outcomes confirmed that these particles had been extraordinarily mild—about 1/2000 the mass of the smallest recognized atom.
From this, Thomson concluded:
Later discoveries of protons and neutrons defined how atoms may include negatively charged electrons whereas remaining electrically impartial general.
Electrons and Atomic Quantity
Though electrons are tiny, they occupy most of an atom’s quantity. The electron cloud surrounding the nucleus is generally empty area, which explains why atoms usually are not stable in the way in which they seem at a macroscopic scale.
Electrons stay close to the nucleus because of the electrical attraction between their destructive cost and the constructive cost of protons.
Why Electrons Are Important
Electrons are answerable for:
-
Chemical bonding
-
Electrical conductivity
-
The properties of parts
-
The formation of molecules
-
Power switch in chemical reactions
With out electrons, atoms couldn’t work together, molecules couldn’t type, and matter as we all know it might not exist.
Conclusion
Electrons are elementary to atomic construction and chemical habits. Their destructive cost, group into power ranges, and function as valence electrons clarify how atoms bond and why parts behave otherwise from each other.
From their discovery in cathode ray tubes to their central function in fashionable chemistry, electrons have reshaped our understanding of matter. Although extremely small, they govern the construction, stability, and variety of the fabric world.

